Example type
Serum, plasma, other biological fluids
Test type
Sandwich (quantitative)
Sensitivity
1.7215 ng / ml
Distance
6.25 ng / ml – 400 ng / ml
Recovery
>85%
Trial duration
Standard multi-step test
Species reactivity
Reacts with: Human
Product description
The Abcam Alpha 1 Antymotrypsin Human ELISA Kit is an in vitro enzyme-linked immunosorbent assay (ELISA) for the quantitative measurement of alpha 1 antitrypsin levels in human serum and plasma.
In this assay, the alpha 1 Antichymotrypsin present in the samples reacts with the anti-alpha 1 Antichymotrypsin antibodies that have been adsorbed on the surface of the polystyrene microtiter wells. After removal of unbound proteins by washing, horseradish peroxidase (HRP) conjugated anti-alpha 1 Antichymotrypsin antibodies are added. These enzyme-labeled antibodies form complexes with previously bound alpha 1 Antichymotrypsin. After another washing step, the enzyme bound to the immunosorbent is analyzed by the addition of a chromogenic substrate, 3,3 ‘, 5,5’-tetramethylbenzidine (TMB).
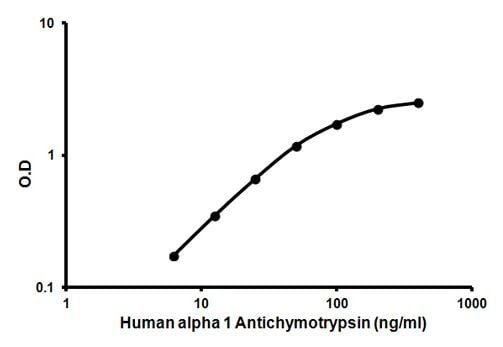
The amount of enzyme-bound varies directly with the concentration of Alpha 1 Antichymotrypsin in the sample tested; therefore, the absorbance, at 450 nm, is a measure of the concentration of alpha 1 Antichymotrypsin in the test sample. The amount of alpha 1 antitrypsin in the test sample can be interpolated from the standard curve constructed from the standards and correct for sample dilution.
Function
Although its physiological role is unclear, it can inhibit neutrophil cathepsin G and mast cell chymase, both of which can convert angiotensin-1 to active angiotensin-2.
Tissue specificity
Plasma, Synthesized in the liver. Like the related alpha-1-antitrypsin, its concentration increases in the acute phase of inflammation or infection. It is found in the amyloid plaques of the hippocampus of the Alzheimer’s disease brain.
Participation in the disease
Defects in SERPINA3 can be a cause of the chronic obstructive pulmonary disease (COPD) [MIM: 107280].
Sequence similarities
It belongs to the Serpin family.
Domain
The reactive center loop (RCL) extends from the body of the protein and directs binding to the target protease. The protease cleaves the serpin at the reactive site within the RCL, establishing a covalent bond between the carboxyl group of the serpin reactive site and the serine hydroxyl of the protease. The resulting inactive serpin-protease complex is very stable.

